 Molecular Models Lab Name: Formula Lewis Structure Electron Domains Geometry Shape of the Bond around molecule angles central atom(s) Lab Day: Polarity of Polar or Hybridization Total Individual non- of the central Number bonds polar? atom of o and bonds N2 N/A N/A 02 N/A N/A N/A N/A Br2 c. Count and state the total number of sigma (o) and pl (n) bonds in the molecule. Determine the hybridization of the central atom. Infer the polarity of the molecule using the shape and bond polarities. There are a total of four electron density regions around the central Cl atom in ClO 4 lewis structure. If there is more than one type of bond, calculate the electronegativity difference for each bond, and state the polarity for each type of bond. The Lewis structure of a perchlorate ClO 4 ion consists of a chlorine (Cl) atom at the center it is bonded to four atoms of oxygen (O) at the sides. The geometry of the regions of electron density is roughly the same as what we see when no NB pairs are involved. Two carbons in the C3H6 molecule forms Sp 2 hybridization and one forms Sp 3 hybridization. The lewis structure of C3H6 has 9 bonding pairs and zero lone pairs. 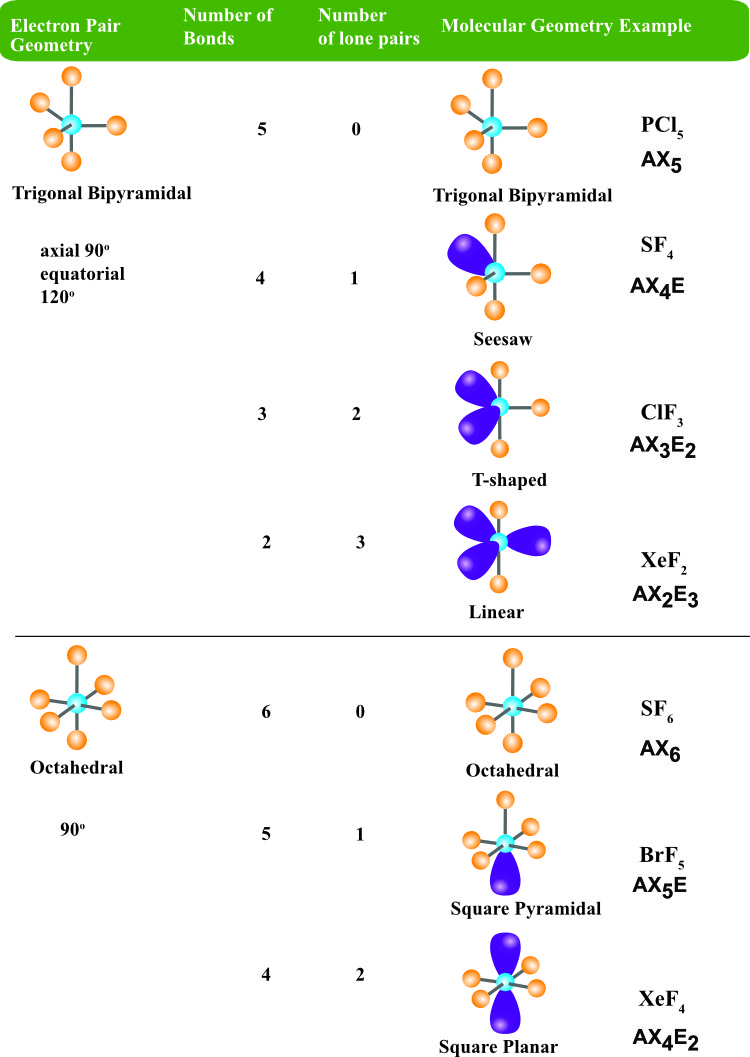
Determine the polarity of all bonds in the molecule. C3H6 has two types of molecular geometry, tetrahedral and trigonal planar. 
Determine the electron pair geometry, molecular shape (also called the molecular geometry), and bond angles in the molecule. Once you build a few of the molecules, have your instructor initial them in your chart. 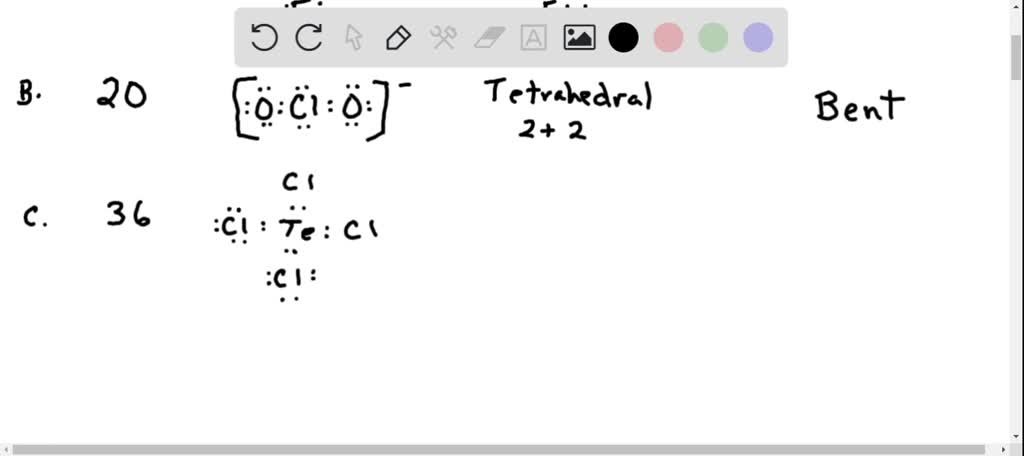
Build a model for each of the atoms in the table below. For each of the molecules in the table, draw the Lewis structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
